What is a chemical reaction?

A chemical reaction involves chemical changes.
What is a chemical change?

A chemical change is where a substance changes into another substance by the reorganization of its atoms.
It's possible to determinate when a chemical change has occurred by looking for some indicators like:
the temperature of the container has changed or the production of a gas or an odor.
It's possible to determinate when a chemical change has occurred by looking for some indicators like:
the temperature of the container has changed or the production of a gas or an odor.
Types of chemical reactions.
Synthesis
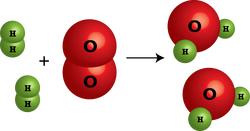
A chemical reaction in which two or more simple compunds combine to form a more complicated one.
Decomposition

A complex molecule breaks down to form a more complicated one.
Single replacement
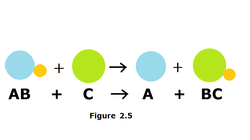
When one element of a reactant trades place with another element of the different reactant to form two new compounds.
Double replacement
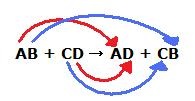
When the anions and cations of two different molecules switch places, forming two entirely different compounds.
Combustion

When oxygen combines with another compound to form water and carbon dioxide.
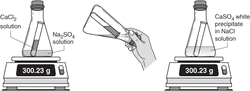
What is a Precipitate?
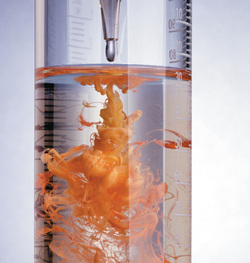
When two liquids are mixed to form a solid.
What is a aqueous solution?

When a solid is dissolved in water.
Law of Conservation of Matter
Matter can neither be created nor destroyed.
Chemical equations
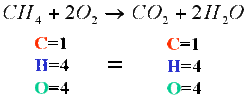
Are used to represent chemical reactions.
